The Climate Energy Balance
Alan K. Betts (akbetts@aol.com)
November 29, 2015 (updated)
The temperature of the Earth is a balance between heating by the sun and the cooling of the Earth to space. As the greenhouse gas CO2 increases, the cooling of the Earth to space is reduced, so the climate is getting warmer. The warming of the Earth is amplified by two things: water vapor in the atmosphere increases with temperature and water vapor is also a strong greenhouse gas; and as more snow and ice melt, the reflection of sunlight is reduced.
The Earth
The Earth’s climate is forced at the top-of-the-atmosphere by the short-wave radiation coming from the sun. Some of this energy (31%) is reflected back to space at once: we call this reflectivity of the whole earth, the planetary albedo. Per unit area, clouds, snow and ice reflect a lot of the sun’s energy (60 - 80%), so any reduction in the fractional coverage of clouds, snow or ice increases significantly how much of the sun’s energy heats the earth and oceans. (See Figure .) Locally in winter, the large reflection of sunlight by snow cover helps lock-in cold winter temperatures (see Seasonal Climate transitions).
The Land:
Vegetation over land has a much lower ‘albedo’ than snow and ice (See Figure .) (10-15% for forests and 15-20% for grasslands), so vegetated surfaces absorb most of the sun’s energy. They use only 1-2% for photosynthesis to grow. A little heat is conducted into the earth in summer, but most of the sun’s energy is used to evaporate water (much as transpiration by plants, which is lost through their leaf pores (stomata) as they take in CO2 for photosynthesis). Some energy is radiated back to the atmosphere as long-wave (thermal or infrared) radiation, and some is lost directly to the air by turbulence and convection. Sandy deserts have little water to evaporate, so they get hotter than vegetated surfaces, despite having a higher surface albedo (25-40%).
The Oceans:
The oceans have a very low albedo (8%), so 92% of the sun’s energy that reaches the surface enters the ocean, and penetrates a few meters until it is absorbed. If there is a lot of plankton, the water is less clear and the sun’s heat does not penetrate as deeply. This heat is then mixed down further by turbulence (driven by the surface waves) into an upper ocean ‘mixed’ layer that may be 50-200m deep. The ocean is cooled mainly by the evaporation of water at the surface that increases as the wind gets stronger or the air gets drier. (See Figure .) Just as over land, there is a long-wave radiative heat loss from the ocean surface; and some heat transfer from water to air, which can be very large if cold, winter air blows off the continent over warm ocean currents like the Gulf-stream. As water evaporates from the ocean, leaving the salts behind, the ocean mixed layer increases in salinity. Over longer time-scales, the ocean circulations transfer heat and salinity from the surface mixed layer down into the deep ocean.
Arctic Sea-ice:
The very large difference between the albedo of the open ocean and of sea-ice plays a critical role in the Earth’s climate. In recent decades, as the Arctic has warmed, the reduction of the Arctic sea-ice in summer (See Figure .) means that the open ocean absorbs more sunlight (less is reflected). This is a positive feedback that has accelerated the warming of the Arctic. There is a second positive feedback here. Water evaporates much more from open ocean than from sea-ice; and the extra water vapor in the atmosphere (a powerful greenhouse gas – see below) reduces the cooling of the Arctic to space. The increase of evaporation from the ocean has a third impact. Storminess increases and stronger winds can blow and breakup more ice. In decades past, the Arctic sea-ice was much thicker multi-year ice (it lasted for several years). Now the sea-ice is thinner and much of it was formed only the previous winter, so it melts more readily in summer (1).
Energy balance of the Earth and the Greenhouse gases:
Solar energy heats the earth, and the earth cools to space by infrared radiation, which increases with temperature. So to reach balance, the earth warms up until it can radiate to space the energy that it gains from the sun. This is where the greenhouse gases (GHGs) are important. The GHGs are the gases that strongly absorb the earth’s thermal infrared radiation, so they effectively blanket the earth (by radiating energy back down to the surface) and reduce the cooling of the earth to space (because the radiation to space comes from higher in the atmosphere, where it is cooler). The primary GHGs are water vapor, carbon dioxide, methane, nitrous oxide, and ozone (H2O, CO2, CH4, N2O, O3), but there are many other industrially produced gases (like the hydrofluorocarbons, HCFCs). These gases are smaller constituents of the atmosphere, but they have modes of rotation and vibration that absorb the earth’s thermal radiation strongly. (Most of the atmosphere is nitrogen and oxygen, but these simple diatomic molecules are too tightly bound to absorb this thermal radiation.). The GHGs are critical to giving the earth the warm climate that supports life – without them the earth on average would be about 33◦C (59◦F) cooler and the oceans would freeze.
The water vapor greenhouse:
Water vapor, which varies strongly across the earth with surface evaporation and atmospheric transports, is the most important GHG. The earth’s water cycle (see discussion paper), is in balance on timescales of a few weeks, so atmospheric water vapor goes up with temperature on these short timescales. Essentially water, evaporated from the surface, rains out in a few weeks and the latent heating from the condensation of water in the atmosphere (which falls out as precipitation) balances the radiative cooling of the atmosphere. This means that the water vapor greenhouse effect has a rather large positive feedback to the climate system temperature, because if temperature rises, more evaporation gives more water vapor in the air, and this traps more of the earth’s thermal radiation.
The CO2 greenhouse:
CO2 is the second most important greenhouse gas and although there is much less CO2 than H2O in the air, it plays a critical role. Atmospheric CO2 is controlled by very different processes than is water vapor, and currently it is not at all in balance. The amount of CO2 has increased 43 % in the past 100 years from about 280 to 400 parts per million (ppm). (See Figure .) There are large exchanges of CO2 between land, atmosphere and oceans. The yearly oscillations are because CO2 goes up in the northern hemisphere winter, when plants respire and release CO2; and down in the summer when photosynthesis takes up CO2. In the natural earth system, before the human impact became large, these were close to being in balance over the global annual cycle. However the input of CO2 from burning fossil fuels and other industrial processes is so large than the biosphere and oceans can only take up about half of the human input – and this fraction appears to be decreasing with time (although with large variability). Consequently atmospheric CO2 is increasing quite rapidly, and the lifetime of this human waste product (coming from primarily the burning of fossil fuel) is a century or more. In fact some is dissolving in the oceans, which are becoming less alkaline, and some of this CO2 will be returned to the atmosphere centuries from now, once we have limited the use of or exhausted our fossil fuel reserves.
The Earth’s Energy Imbalance:
Because of increased GHGs in the atmosphere, the Earth at present has a net energy gain2 of about 1-2 Wm-2. A small part of this energy imbalance is melting ice, but the Earth must get warmer on average, until this imbalance can be radiated back to space. This warming of the Earth has a lag of 50-100 years because of the large thermal storage of the oceans. Furthermore, the climate system has instabilities and feedbacks2 that are only partially understood, so there are large uncertainties in our projections for the future climate of the Earth.
The Ice-age message:
We know from the ice-age records of the past million years that there are some strong positive feed-backs in the Earth system. On time-scales between forty and a hundred thousand years, changes in the inclination of the Earth’s axis and the geometry of the Earth’s orbit around the sun gave small changes in the solar radiation reaching the earth (15-20% at 60◦N in June). These small radiative changes were amplified by the climate system to give changes of temperature at the poles of order 10◦C (18◦F). During glacial maxima, ice-sheets grew to more than double their present size, reflecting much more sunlight, and lowering sea-level by as much as 120 m (nearly 300ft). (See Figure .)
Age-age cycles appear to have ended during periods of higher solar radiation in summer at northern latitudes; with ice-sheet collapse taking only a few thousand years. On ice-age time-scales, as temperature fell, CO2 fell from 280 to 180 ppm, with the weathering of rocks and transports of carbon into the deep ocean; and methane fell from 700 to 350 ppb. Solar forcing was the driver, but the three GHGs (H2O, CO2, CH4) greatly amplified the climate change.
However, the anthropogenic change in atmospheric CO2 that is driving the present climate shift is much faster than any of the natural Earth system processes seen in the ice-age cycles. We are seeing a large amplification, as declining ice cover reflects less sunlight, and the increasing water vapor greenhouse traps more of the earth’s long-wave heat radiation; but the Earth’s temperature and atmospheric CO2 are much higher than during the ice-ages, so it is hard to predict exactly what will happen to the Earth. It has been millions of years since the Earth’s atmosphere had 400-500 ppm CO2 (which we are again approaching), and then the Earth may have had no ice-caps and a very different climate and vegetation – long before humanity evolved. This is why Hansen (2) has suggested that atmospheric CO2 should be brought back down to 350 ppm as soon as possible.
Other Arctic Feedbacks:
There are some other processes that are accelerating the warming of the Arctic. Soot from airborne pollution darkens the ice (3), which reduces the albedo and accelerates melting. As the Arctic warms, shrubs are growing in the tundra regions, and this also darkens the surface and accelerates the warming We suspect other processes will become important as the Arctic warms. Permafrost is thawing, and this could lead to accelerated production of methane from the anaerobic decomposition of the deep layers of peat that have been frozen for millennia. In the oceans there are large stores of methane hydrates that could also be released as the ocean warms.
- Kwok, R. and D. A. Rothrock, 2009: Decline in Arctic sea ice thickness from submarine and ICESat records: 1958–2008. Geophys. Res. Lett., 36, L15501, 5 pp., doi:10.1029/2009GL039035
- Hansen, J. Et al. (2008), Target Atmospheric CO2: Where Should Humanity Aim? The Open Atmospheric Science Journal, 2, 217-231.
- Hansen, J. and L. Nazarenko (2004), Soot climate forcing via snow and ice albedos. PNAS, 101, 423-428 doi: 10.1073/pnas.2237157100.
Figures and Images
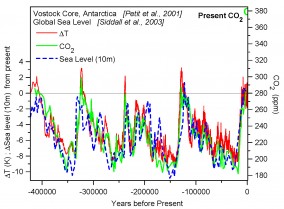 Figure 1: Ice-ages changes of temperature, CO2 and sea-levelChanges through the ice-ages of polar temperature, atmospheric CO2 and sea-level (multiply by 10 for m and 33 for ft)
Figure 1: Ice-ages changes of temperature, CO2 and sea-levelChanges through the ice-ages of polar temperature, atmospheric CO2 and sea-level (multiply by 10 for m and 33 for ft)![Increase of atmospheric CO2 [Credit: NOAA/ESRL]](https://alanbetts.com/image/1/284/0/uploads/co2_data_mlo2015-565b4b87ad7eb.jpg) Figure 2: Increase of atmospheric CO2 [Credit: NOAA/ESRL]Increase of atmospheric CO2 from 1958-2008 at Hawaiian observatory.
Figure 2: Increase of atmospheric CO2 [Credit: NOAA/ESRL]Increase of atmospheric CO2 from 1958-2008 at Hawaiian observatory.![Declining Arctic sea-ice cover [Credit: National Snow and Ice Data Center]](https://alanbetts.com/image/1/284/0/uploads/arcticseaice-sept2015-565b4b5bd8381.jpg) Figure 3: Declining Arctic sea-ice cover [Credit: National Snow and Ice Data Center]September sea-ice extent from 1979 to 2012 shows a continued decline. The September rate of sea ice decline since 1979 has now increased to 13% per decade.
Figure 3: Declining Arctic sea-ice cover [Credit: National Snow and Ice Data Center]September sea-ice extent from 1979 to 2012 shows a continued decline. The September rate of sea ice decline since 1979 has now increased to 13% per decade. Figure 4: Oceans and cloudsSunlight heats the ocean, evaporation cools it, clouds form and reflect some sunlight
Figure 4: Oceans and cloudsSunlight heats the ocean, evaporation cools it, clouds form and reflect some sunlight Figure 5: Winter reflection by snow, Pittsford, VermontSnow reflects sunlight except where shaded by trees- so forests in the background look dark
Figure 5: Winter reflection by snow, Pittsford, VermontSnow reflects sunlight except where shaded by trees- so forests in the background look dark Figure 6: The sun heats the Earth: clouds and ice reflect sunlightThe sun heats the Earth: clouds and ice reflect sunlight
Figure 6: The sun heats the Earth: clouds and ice reflect sunlightThe sun heats the Earth: clouds and ice reflect sunlight